Which of the Following Statements About Subatomic Particles Is True
The mass of a proton is similar to that of a neutron D. The true statement about subatomic particles is that Electrons are the subatomic particles with the smallest mass.

Solar Nebula Theory Solar Nebula Theories Class Notes
Unlike protons and electrons neutrons have no charge.

. The statement which is true about sub atomic particles is electrons are the subatomic particles with the smallest mass. A neutral atom always has the same number of neutrons and electrons. Protons and neutrons have charges of opposite signs but the same magnitude.
Which of the following statements about subatomic particles are true. Electrons have very little mass compared to protons and neutrons. Which of the following statements about subatomic particles is true.
The force between two electrons is equal in magnitude and same direction as that between a proton and an electron. B For a neutral atom the atomic number is equal to the proton number. A protons weigh more than electrons and neutrons b protons weigh Subjects.
Which statement about subatomic particles is true. Charge of proton Neutrons have no chargeAn atom is composed. The 3 subatomic particles share key differences and similarities in their masses and charges.
Which of the following statements about subatomic particles are true. A neutral atom contains the same number of protons and electrons. This is a true statement because opposite charges attract each other whereas like charges repel each other.
A Protons neutrons and electrons are the three subatomic particles present in an atom. Protons are positively charged and the lightest subatomic particle. Protons and neutrons are present inside the nucleus of atom and electrons are present outside the nucleus.
Which of the following statements about subatomic particles is TRUE. Which of the following statements about the masses of subatomic particles is true. Electrons have very little mass compared to protons and neutrons.
The mass of a neutron nearly equals the mass of a proton. Which statement is true about electrostatic force between subatomic particles. Label each type as put down reasoning rejection or Acid precipitation lowered the ph of soil in a terrestrial ecosystem that supported a diverse community of plants and animals.
Which of the following statements about the masses of subatomic particles is true. Protons and neutrons have charges of opposite signs but the same magnitude. Of 11840 of an atomic mass unit amu.
Protons and neutrons have about the same mass. Atoms is known to have three basic particles which are protons electrons and neutrons. Osmium one of the densest elements on earth has an actual mass of 19023 grams.
Which of the following statements about subatomic particles are true. B A neutral atom contains the same number of protons and electrons. Select all that apply A.
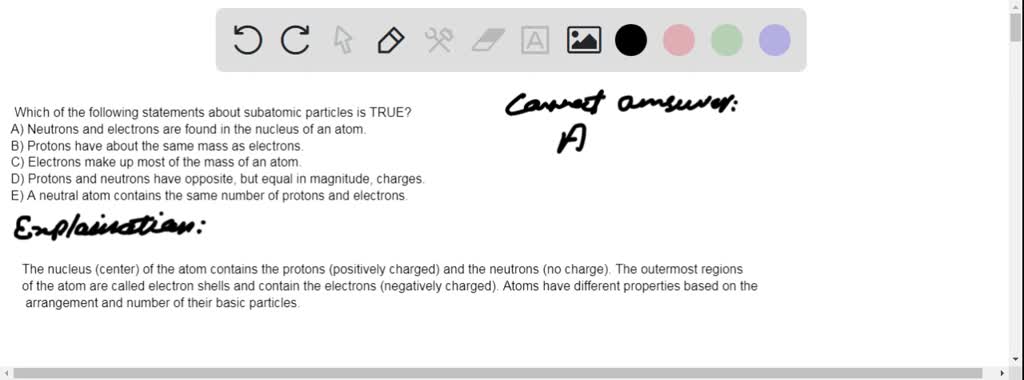
A neutral atom always has the same number of neutrons and electrons. Which of the following statements about subatomic particles is true AnswerProtons have about the same mass as electrons is falseExplanationElectrons and protons have the same magnitude on charge but opposite sign. Select ALL that apply the particles will have the same kinetic energy the space between the particles is different with.
Which statement about subatomic particles is NOT true. Charge of electron. Protons and neutrons have about the same mass.
1 point the number of neutrons the charge on the. Which of the following is unique for any given element. The force between two electrons is equal in magnitude and opposite in direction as that between an electron and a proton.
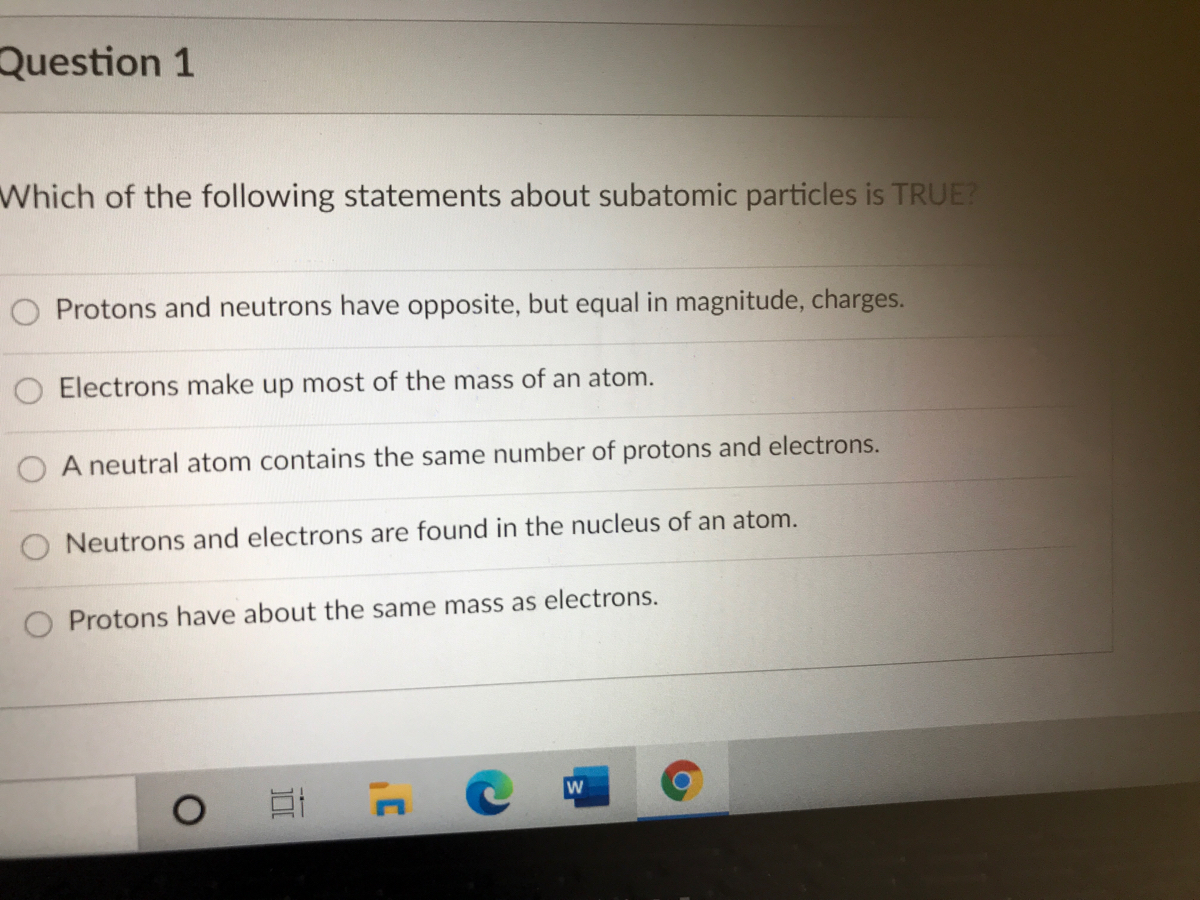
The charge on a proton is equal in magnitude and opposite in sign to that of an electron C. Correct answer - Which of the following statements about subatomic particles is TRUE. O Neutrons and electrons are found in the nucleus of an atom.
Which subatomic particles are found in the nucleus of an atom of beryllium. Electrons are negatively charged and are the heaviest subatomic particle. According to the table above what is its value in terms of atomic mass units.
Which of the following statements about subatomic particles are correct. Which statement is true about the particles in liquid water at 100ºC and the particles in steam at 100ºC. Which one of the following statements about atoms and subatomic particles is correct.
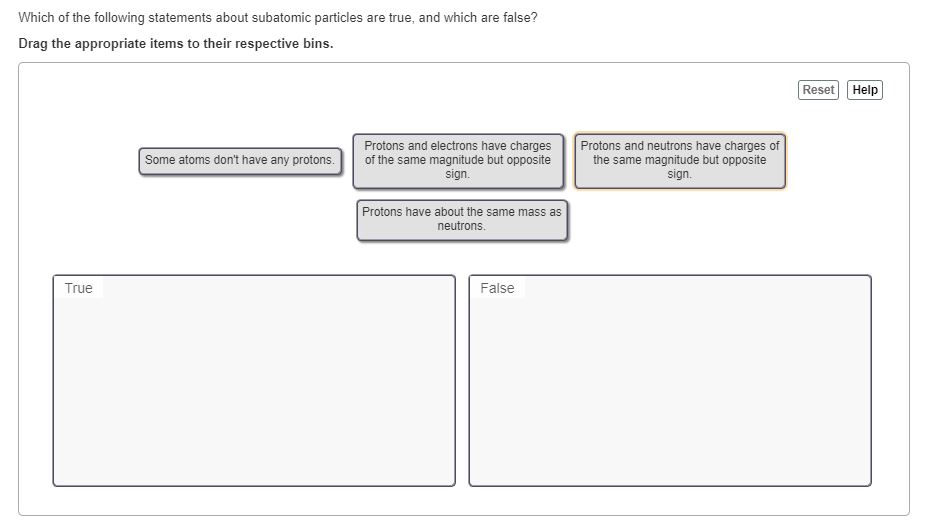
Neutrons and protons are the only charges subatomic particles B. Protons have twice the mass of neutrons. Protons and electrons have charges of opposite signs but the same magnitude.
Neutrons have no charge and are the lightest subatomic particle. Electrons are lighter than neutrons. This is a true statement because the charge of the proton and the electron are equal in magnitude but opposite in sign so that when the two particles are paired the charges sum to zero.
Electrons are the subatomic particles with the smallest mass since it has a mass of 91094 x 10 31 kg while protons has a mass of 16726 x 10-27 kg. O Electrons make up most of the mass of an atom. Protons and neutrons have very little mass compared to electrons- wrong Electrons have no mass.
Which statement describes the location of two types of. There are subatomic particles the electron is known to have the smallest mass. Protons and neutrons have the same charge.
Protons neutrons and electrons all have about the same mass. Protons and electrons have opposite charges. O Neutrons and electrons are found in the nucleus of an atom.
O Electrons make up most of the mass of an atom. O Protons have about the same mass as electrons. Protons neutrons and electrons all have about the same mass.
1 point Protons and neutrons have almost the same mass. Think about which type of peer pressure the statements fall into. Which subatomic particles contribute to the mass of an atom.
O A neutral atom contains the same number of protons and electrons. Which of the following statements about subatomic particles is TRUE. Protons and neutrons have opposite but equal in magnitude charges.
Which of the following statements about subatomic particles is TRUE A Protons from SOC 105 at University of Michigan. Which of the following is true about subatomic particles. Neutron has a mass of 16741 x10-27 kg.

Atomic Theory Worksheet Answers Atomic Theory Word Problem Worksheets Education Templates

Where Do The Laws Of Physics Come From Physics Visual Dictionary Understanding

Chapter 2 Matter And Atomic Structure Edited

Which Statement About Subatomic Particles Is True Youtube
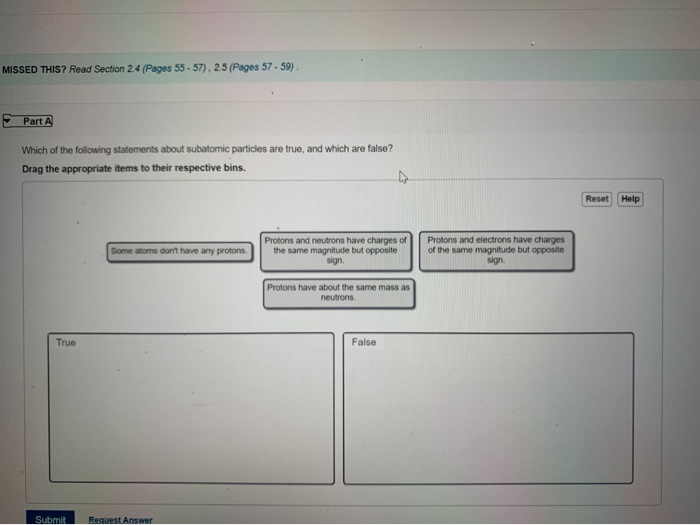
Solved Missed This Read Section 2 4 Pages 55 57 2 5 Chegg Com

Solved Which Of The Following Statements About Subatomic Chegg Com

What Is True About Subatomic Particles
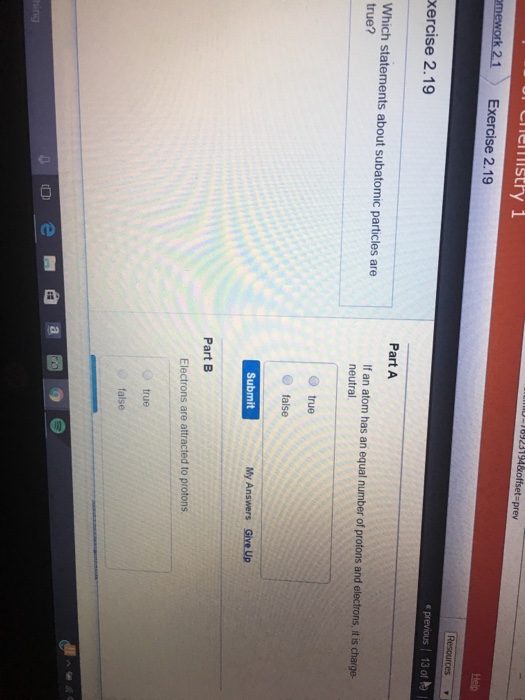
Solved Which Statements About Subatomic Particles Are True Chegg Com

Solved Which Of The Following Statements About Subatomic Particles Are False L Neutrons Reside Inside Of The Nucleus The Atomic Number Is Equal To The Number Of Neutrons In The Nucleus Of An
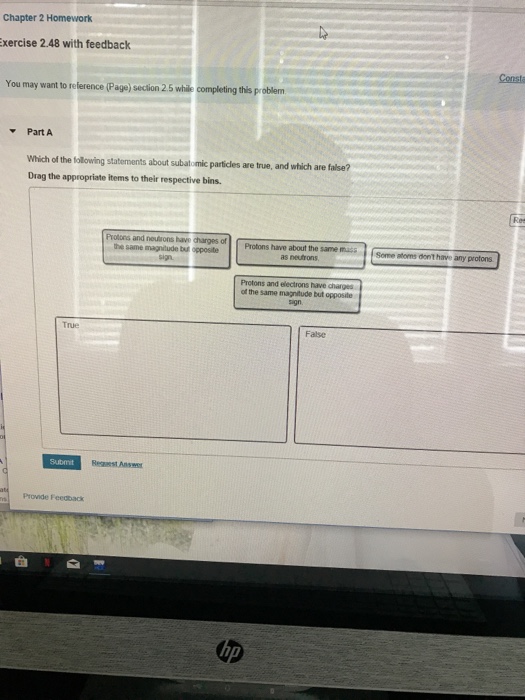
Solved Chapter 2 Homework Xercise 2 48 With Feedback Consta Chegg Com
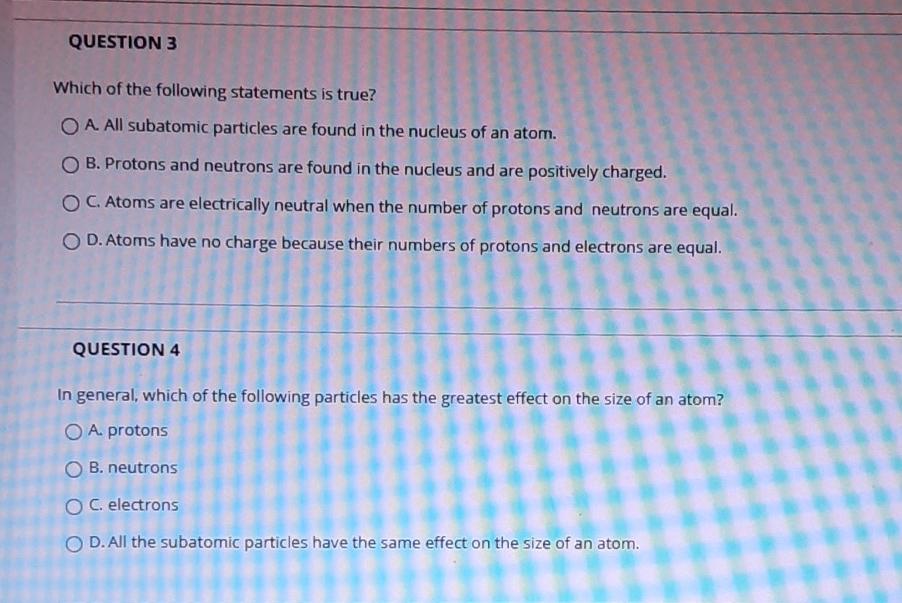
Solved Question 3 Which Of The Following Statements Is True Chegg Com

Solved Three Atoms Have The Following Subatomic Particles Atom 1 12 Protons 6 Neutrons 12 Electrons Atom 2 8 Protons 10 Neutrons 8 Electrons Atom 3 Protons 9 Neutrons Electrons Which Of The
Answered Which Of The Following Statements About Bartleby
Solved 5 Which Of The Following Statements About Subatomic Particles Is True A Neutrons And Electrons Are Found In The Nucleus Of An Atom B Protons Have About The Same Mass As Electrons

Pin By Sharon Strangman On Motion Views Optical Illusion Gif Illusion Gif Cool Optical Illusions

Solved Which One Of The Following Statements About Atoms And Chegg Com
Answered Which Of The Following Statements About Bartleby

Solved Which Of The Following Statements About Subatomic Chegg Com

Comments
Post a Comment